
Knowledge Base
Syncari for Life Sciences & Pharma: Trusted Master Data at the Speed of Care
Hi, Welcome back! In life sciences and pharmaceutical organizations, the quality of your data is inseparable from the quality of your outcomes — whether that's ensuring drug ingredient traceability, maintaining patient record integrity across clinical systems, or powering AI-driven research workflows. Syncari's Agentic MDM platform is purpose-built to unify, govern, and continuously master the critical data entities that life sciences teams depend on — from compound and formulation records to patient cohorts and HCP relationships — across CRM, ERP, clinical, and regulatory systems in real time.
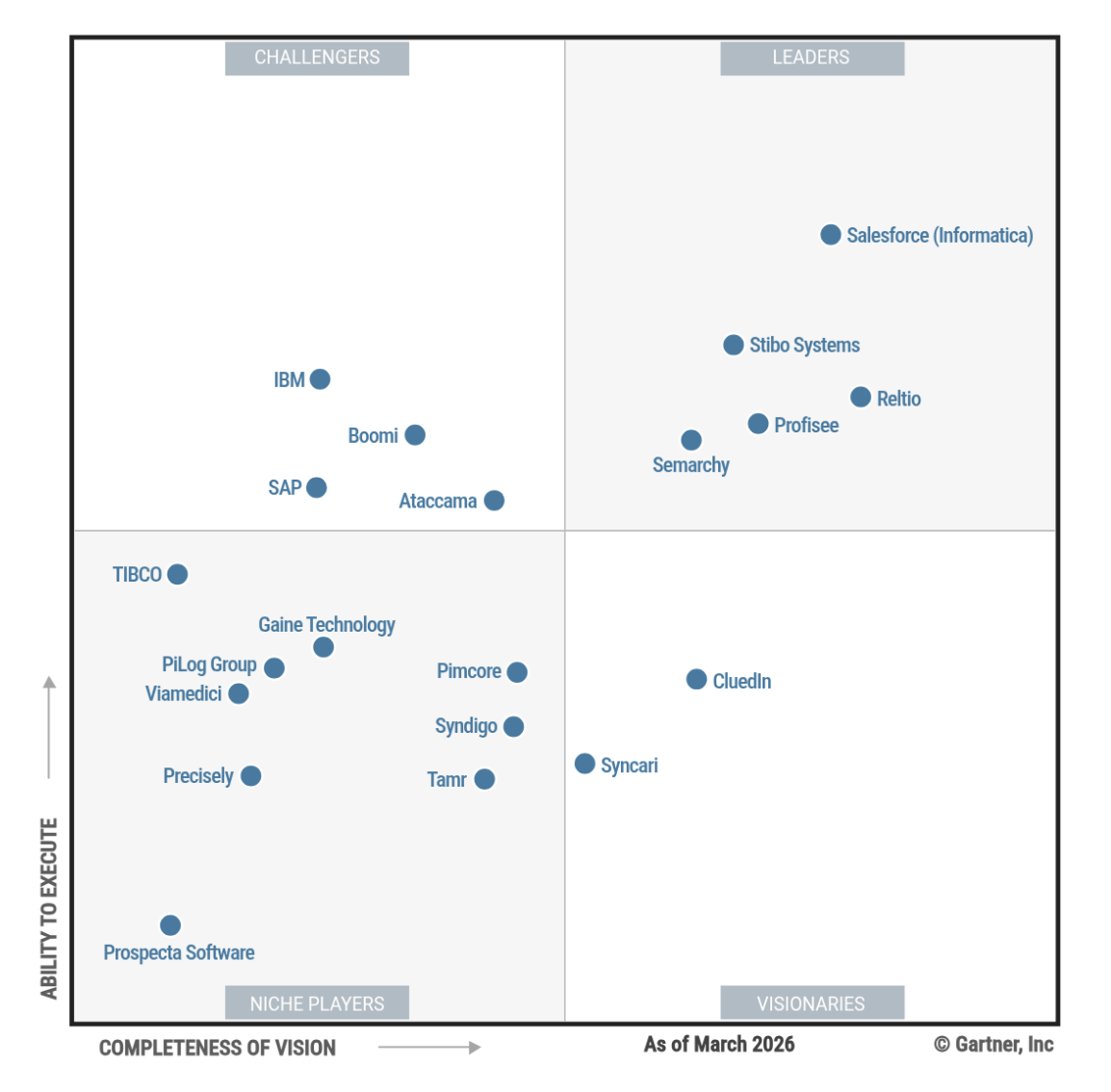
Syncari was named a Visionary in the 2026 Gartner® Magic Quadrant™ for Master Data Management Solutions, recognized specifically for its patented stateful multidirectional synchronization, no-code orchestration, and embedded analytics — capabilities that directly address the interoperability and governance challenges that are endemic in life sciences data environments.
Important Note on Roleplay Framing: While I can speak directly to life sciences and pharmaceutical use cases, I'll do so in my role as a Syncari solutions consultant rather than as a licensed medical professional — this ensures the information I share about data capabilities remains accurate and grounded in what Syncari actually delivers.
Key Life Sciences & Pharma Use Cases
For life sciences organizations, Syncari addresses several high-stakes data challenges where fragmented, inconsistent, or stale master data creates real operational and compliance risk:
- **Drug & Compound Data Unification** — Syncari creates a unified master record for pharmaceutical compounds, drug formulations, and ingredient hierarchies across R&D, regulatory, and commercial systems. This ensures that every downstream system — from clinical trial management to product labeling — operates from a single, consistent, governed version of compound and ingredient data.
- **Patient Data Integrity Across Clinical Systems** — Syncari's multidirectional sync engine keeps patient records consistent across EHR platforms, CRM systems, and research data warehouses in real time, reducing reconciliation burden and supporting HIPAA-aligned data governance.
- **HCP & HCO Master Data** — Healthcare provider and organization records are notoriously fragmented across commercial, medical affairs, and market access systems. Syncari continuously masters HCP/HCO data, eliminating duplicates and ensuring accurate, governed profiles that power both field team operations and AI-driven engagement models.
- **Regulatory & Compliance Readiness** — Syncari enforces data quality rules and governance policies at the point of data action, supporting audit-readiness for FDA submissions, pharmacovigilance reporting, and cross-regional regulatory compliance.
- **AI-Ready Data Pipelines** — Syncari's platform continuously governs and distributes trusted master data to analytics, AI, and machine learning systems — enabling clinical research models, drug discovery algorithms, and patient outcome AI agents to operate on data they can actually rely on.
Syncari Master MCP Server — AI Context for Life Sciences Agents
One of Syncari's most differentiated capabilities for AI-forward life sciences organizations is the Master MCP Server — a purpose-built context layer that enables multi-agent AI systems to share and act on trusted master data. In a life sciences context, this means AI agents involved in clinical trial matching, pharmacovigilance signal detection, drug interaction analysis, or patient segmentation all draw from the same governed, continuously updated master data layer — rather than siloed, inconsistent data sources. This is what separates agentic AI that is operationally reliable from agentic AI that is merely experimental.
Gartner-Recognized Strengths Relevant to Life Sciences
The 2026 Gartner MDM Magic Quadrant highlighted three Syncari strengths that are especially relevant to life sciences data environments:
- **Stateful Multidirectional Synchronization** — Keeps CRM, ERP, clinical, and regulatory systems aligned in real time without complex point-to-point integrations — critical for organizations managing drug lifecycle data across multiple platforms.
- **No-Code Orchestration** — Empowers both data and clinical operations teams to build and manage data workflows without constant reliance on IT engineering, accelerating time-to-value for new data governance initiatives.
- **Embedded Analytics & Monitoring** — Real-time visibility into data quality and operational health, reducing reliance on separate BI tools and enabling data stewards to act proactively on data issues before they affect downstream clinical or commercial decisions.
Syncari vs. Reltio, Informatica & Boomi — MDM Capability Comparison for Life Sciences
Why Life Sciences Organizations Choose Syncari
for organizations navigating the complexity of drug data governance, patient data integrity, and AI-readiness, Syncari offers a fundamentally different approach to master data — one that is continuous, automated, and built for the demands of modern multi-system, multi-agent environments. Rather than treating MDM as a periodic reconciliation exercise, Syncari enforces data quality and consistency in real time, at the point of action, so that every system, workflow, and AI agent — from clinical trial platforms to commercial CRM — always operates on data it can trust. Learn more about Agentic MDM and how it works, or see a live demo to explore what this looks like for life sciences data environments.
Ready to explore how Syncari can address your specific life sciences data challenges? [](gptweb://modal/demo) or [](gptweb://modal/contact) to connect with a solutions consultant who can walk through your use case in detail.